The migratory primary mesenchyme cells showed just the opposite behavior. Results showed that pre-migratory micromeres adhered 100 times stronger to the hyaline layer and other blastomeres than to the basal lamina. They then added 16 cell stage micromeres, migratory stage primary mesenchyme cells, gastrula ectoderm and endoderm. They first made test plates to which they bound hyaline, test cell monolayers, or basal lamina. This model that suggests differing adhesive interactions is responsible for the ingression of the primary mesenchyme was directly tested by Fink and McClay. These cells first migrate randomly in the blastocoel, but soon become localized on the ventral surface of the blastocoel, where they fuse to form the syncytial cables that will give rise to the calcium carbonate spicules. They are fated to give rise to the SKELETAL RODS of the PLUTEUS LAVA. Cells which are the first to INGRESS are called the PRIMARY MESENCHYME and are derived from the MICROMERES. The cells INGRESS as individuals and not as a sheet (not epiboly), not an INVAGINATION, not an INVOLUTION, not a DELAMINATION. The cells loose their adhesion with the surrounding cells of the blastoderm and are pulled inward into the blastocoel by their filopodial processes. They are also attached at their basal surfaces to the basal lamina.Ĭentrally located cells begin to extend filopodial processes from their inner surface. The blastomeres adhere strongly to the hyaline layer and to each other. The blastula is surrounded by the hyaline layer and the blastomeres have secreted a basal lamina surrounding the blastocoel. About 24 hrs after hatching of the ciliated blastula the vegetal side of the blastula begins to flatten to form the VEGETAL PLATE.
#DORSAL LIP OF BLASTOPORE MOVIE#
The fate of blastomeres in the hatched blastula can be easily recognized as arising from the early pattern of cleavages and suggests little cell movement during the blastula stage.Įxcellent movie of sea urchin gastrulation from Rachel Fink's "A Dozen Eggs". Remember at the 4th cleavage the animal pole mesomeres divide in the meridinal plane while the vegetal pole cells divide in the equatorial plane asymmetrically to give rise to the 4 macromeres and the 4 micromeres. Even so, blastomeres do not always cleave symmetrically. The egg is isolecithal and cleavage symmetry is radial holoblastic. Remember the cleavage pattern of sea urchins. The same blastomeres end up with the same egg cytoplasm and in the same positions in the blastuala.
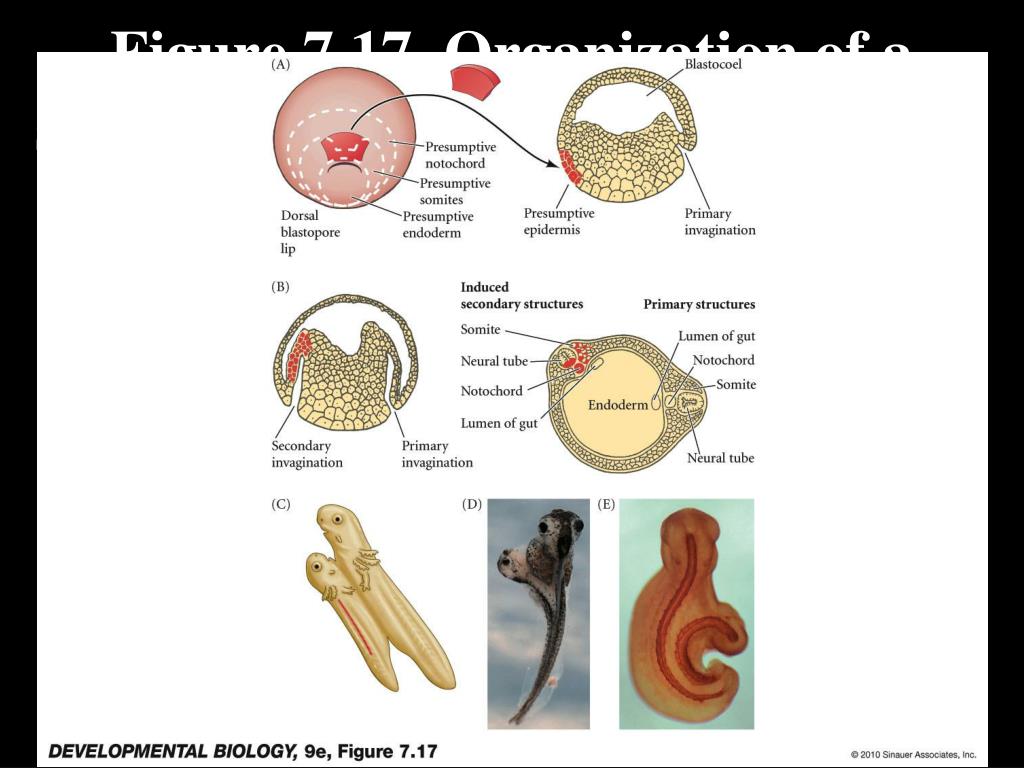
Cells can migrate as individuals, eg., ingression, or as part of a unit with other cells, eg., invagination, involution, delamination, and epiboly.Ī fate map can be drawn on the egg because of the stereotyped pattern of cleavages. New positions and neighbor relationships are determined by the pattern of cell movements at gastrulation. The initial positions and neighbors of the blastomeres is determined by the pattern of cleavages. Development 114, 403–415.Gastrulation is the process of cell movements that give rise to the primary germ layers of the embryo. (1992) Commitment of mesoderm cells in Hensen’s node of the chick embryo to notochord and somite. IRL at Oxford University Press, Oxford, UK. (eds.) (1993) Essential Developmental Biology: A Practical Approach. H., eds.), IRL at Oxford University Press, Oxford, UK, pp. (1993) Transplantation in avian embryos, in: Essential Developmental Biology: A Practical Approach. (1993) Avian embryos, in: Essential Developmental Biology: A Practical Approach (Stern, C. (1981) An integrated experimental study of endoderm formation in avian embryos. (1950) The effects of antero-posterior reversal of lengths of the primitive streak in the chick. (1947) A simple method for explanting and cultivating early chick embryos in vitro. (1995) A role for HGF/SF in neural induction and its expression in Hensen’s node during gastrulation. W., Aparicio, S., Sharpe, M., and Gherardi, E. (1994) The avian embryo: a powerful model system for studying neural induction.

(1992) Neural induction and regionalisation in the chick embryo. (1995) Induction by different subpopulations of cells in Hensen’s node. (1991) Fate mapping and cell lineage analysis of Hensen’s node in the chick embryo. (1955) A new technique for the cultivation of the chick embryo in vitro.
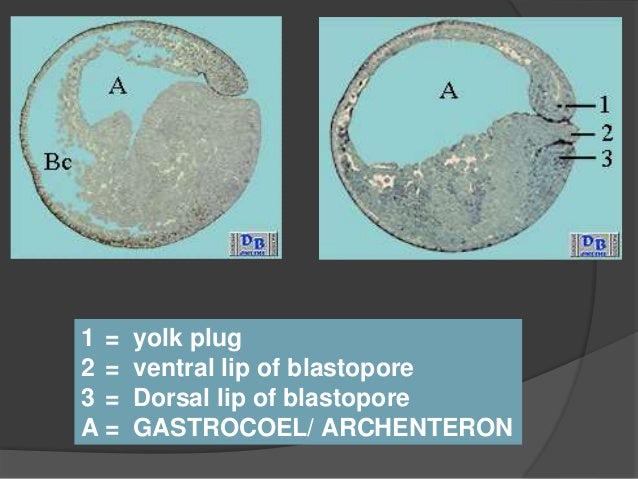
#DORSAL LIP OF BLASTOPORE SERIES#
(1951) A series of normal stages in the development of the chick embryo. (1933) Induction by the primitive streak and its derivatives in the chick. (1932) Experiments on the development of chick and duck embryos, cultivated in vitro. (1924) Über Induktion von Embryonanlagen durch Implantation artfremder Organisatoren.
